For much of the last century, weight management has been framed as a simple arithmetic problem. Calories consumed versus calories expended became the dominant model used to explain weight gain, weight loss, and long-term body composition. This framework shaped public health guidance, diet programs, and clinical interventions, all centered on one primary strategy: eat less.
Yet despite decades of calorie-focused advice, rates of obesity and metabolic disease have continued to rise. Many individuals successfully lose weight through calorie restriction, only to regain it months or years later. Others find that reducing calories produces fatigue, hunger, and metabolic slowdown without meaningful or lasting results. These outcomes have prompted researchers to reexamine the calorie restriction limits of this approach, particularly in the context of metabolic dysfunction.
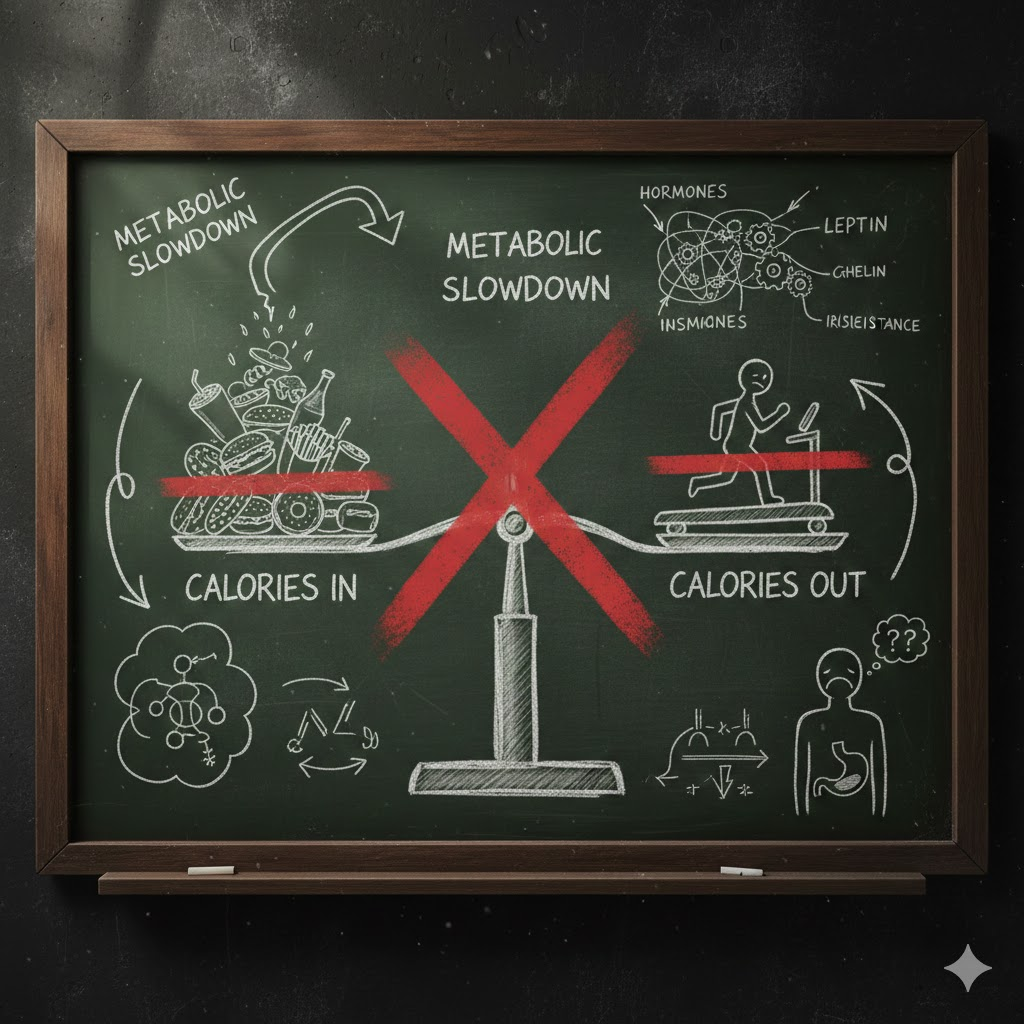
Increasingly, weight science suggests that calorie restriction alone fails not because people lack discipline, but because metabolism adapts in ways that undermine long-term success.
The Origins of the Calorie Model
The calorie model emerged from early studies of energy balance and thermodynamics. In controlled environments, reducing caloric intake leads to weight loss, while excess intake leads to weight gain. These findings were accurate within their experimental context.
However, translating short-term laboratory results into long-term human behavior proved more complex. Humans are not closed systems. Hormones, neural signaling, energy expenditure, and adaptive responses all influence how calories are processed and stored.
As research expanded, it became clear that calories are not metabolically equivalent in how they affect hunger, fat storage, and energy regulation. The body responds to calorie reduction not as a neutral subtraction, but as a biological signal that triggers compensatory mechanisms.
Metabolic Adaptation to Calorie Restriction
One of the most well-documented responses to calorie restriction is metabolic adaptation. When caloric intake drops significantly, the body reduces energy expenditure to conserve fuel. Resting metabolic rate declines, spontaneous movement decreases, and thermogenesis becomes more efficient.
This response evolved as a survival mechanism during periods of food scarcity. In modern contexts, it creates a physiological barrier to sustained weight loss. As calorie intake remains low, weight loss slows or stalls, even when adherence remains high.
Research summarized by the National Institutes of Health has shown that metabolic rate suppression can persist long after calorie restriction ends, increasing the likelihood of weight regain. This adaptation highlights a key limitation of calorie-only strategies.
Hormonal Responses That Undermine Restriction
Calorie restriction also alters hormonal signaling in ways that increase hunger and reduce satiety. Leptin, a hormone that signals energy sufficiency, declines with weight loss and reduced calorie intake. Ghrelin, a hormone that stimulates appetite, increases.
These hormonal shifts are not temporary inconveniences. They represent the body’s attempt to restore energy balance by increasing food intake and reducing expenditure. As restriction continues, hunger signals intensify, making long-term adherence increasingly difficult.
Importantly, these responses occur regardless of willpower. They are biologically driven, reinforcing the idea that calorie restriction limits are rooted in physiology rather than behavior.
Insulin Resistance Changes the Equation
In metabolically healthy individuals, calorie restriction can produce predictable weight loss. In individuals with insulin resistance, the response is often different.
Insulin resistance alters how energy is stored and released. When insulin levels remain elevated, fat mobilization becomes more difficult, even under caloric deficit. The body may reduce muscle mass and metabolic rate while preserving fat stores.
This helps explain why calorie restriction alone often produces disappointing results in individuals with metabolic dysfunction. Weight loss may occur initially, but it disproportionately affects lean tissue rather than fat mass, worsening metabolic efficiency over time.
The American Journal of Clinical Nutrition has published analyses showing that insulin resistance predicts poorer outcomes in calorie-restricted weight loss interventions, reinforcing the need to address metabolic context rather than calories alone.
The Quality of Calories Matters
Another limitation of calorie-only approaches is that they ignore dietary composition. Calories derived from refined carbohydrates, sugars, and ultra-processed foods produce different hormonal and metabolic effects than calories from whole, nutrient-dense foods.
Highly refined foods promote rapid glucose absorption and repeated insulin spikes. These responses drive hunger, fat storage, and energy crashes, making sustained restriction more difficult. In contrast, diets that stabilize blood sugar and insulin signaling often reduce hunger spontaneously, even without intentional calorie reduction.
This distinction helps explain why individuals consuming the same number of calories can experience very different weight and metabolic outcomes.
Psychological and Behavioral Costs
Beyond physiology, calorie restriction imposes psychological costs that undermine long-term success. Chronic hunger, food preoccupation, and reduced energy affect mood, concentration, and motivation.
Over time, restriction can increase stress hormones such as cortisol, which further impair insulin sensitivity and promote fat storage. This creates a paradoxical situation where attempting to lose weight through restriction intensifies the very mechanisms that resist weight loss.
Weight science increasingly recognizes that sustainable interventions must reduce physiological stress rather than amplify it.
Why Weight Loss Plateaus Are Predictable
Weight loss plateaus are often framed as personal failure. From a metabolic perspective, they are expected outcomes of prolonged calorie restriction.
As energy intake drops, the body recalibrates its baseline. Hormonal changes increase hunger, metabolic rate declines, and energy efficiency improves. Eventually, the reduced intake matches the reduced expenditure, halting further loss.
Pushing past this plateau through additional restriction often worsens metabolic adaptation rather than overcoming it. This dynamic illustrates a fundamental calorie restriction limit in the context of long-term weight regulation.
A Broader View of Weight Science
Modern weight science is shifting away from calorie arithmetic toward systems-based models. These models consider insulin signaling, metabolic flexibility, circadian rhythms, stress response, and nutrient availability as interconnected regulators of body weight.
Within this framework, calories still matter, but they are not the primary driver. The body’s hormonal environment determines how calories are partitioned between storage and use. Restriction without addressing that environment produces predictable resistance.
Educational resources focused on metabolic health, including those available on Dr. Berg, often emphasize restoring metabolic signaling rather than enforcing chronic calorie deficits.
Why Calorie Restriction Works Short Term but Fails Long Term
Short-term calorie restriction works because it creates an immediate energy gap. Long-term failure occurs because the body adapts to close that gap.
This distinction explains why calorie-based diets consistently produce early success followed by plateau and regain. The body’s priority is survival, not adherence to dietary plans.
Understanding this pattern reframes weight regain not as relapse, but as a predictable biological response to sustained restriction.
Toward Metabolic-Centered Strategies
Weight science increasingly supports approaches that reduce hunger, improve insulin sensitivity, and support metabolic flexibility rather than relying solely on calorie reduction.
These strategies often result in spontaneous calorie reduction without deliberate restriction, because the body no longer perceives energy scarcity. Hunger decreases, energy improves, and adherence becomes less effortful.
This shift does not reject energy balance, but it recognizes that the body regulates energy through hormones and signaling pathways rather than conscious math.
Rethinking Failure in Weight Management
Labeling calorie restriction failure as a lack of discipline obscures the underlying biology. When interventions repeatedly fail across populations, the model deserves reevaluation.
Recognizing calorie restriction limits allows weight science to move beyond blame-based frameworks and toward approaches aligned with human physiology.
This reframing is particularly important in clinical and public health settings, where ineffective guidance carries long-term consequences.
Calorie restriction alone fails in metabolic dysfunction because it ignores how the body adapts to perceived scarcity. Hormonal changes, metabolic slowdown, and increased hunger are not side effects. They are core features of the response.
Understanding calorie restriction limits provides a clearer explanation for why weight loss is difficult to sustain and why metabolic health must be addressed alongside energy intake.
As weight science continues to evolve, successful strategies are likely to focus less on eating less and more on restoring the metabolic conditions under which the body can regulate weight without constant resistance.
